
Sickle cell disease is the world’s most common inherited blood disorder. The majority of the 5,000 to 6,000 people in Canada affected by this disorder are of African ancestry.
A multidisciplinary research team at Toronto General Hospital’s Red Blood Cell Disorders Program aims to change the lives of people living with sickle cell disease. The team is co-led by Princess Margaret Cancer Centre Clinician Investigators Dr. Kevin Kuo (research lead) and Dr. Richard Ward (clinical lead).
“Our patients are from communities that have historically been marginalized within our society and medical research,” explains Dr. Kuo. “Through close collaboration with our patients as partners, this research is leading to new therapies and models of care for sickle cell disease.”
“Our clinical trials are not only important for advancing research, but also for building meaningful relationships with our study participants,” says Pearl Onugha, Registered Nurse and Clinical Coordinator for the team. “As research health care providers, we find joy in knowing that we are offering novel therapies that can help improve our patients’ care and experience.”
Sickle cell disease is an inherited disorder that causes crescent-shaped red blood cells, which can block blood flow and prevent oxygen from reaching vital organs. Obstructed blood vessels cause immense pain, and over time, patients experience complications such as anemia, stroke and early death. Despite the impact sickle cell disease has on an individual’s quality of life, few reliable treatments are available.
The research group is conducting close to 20 different clinical trials. Combined, these trials touch on the entire health care and treatment journey. Below are some examples of the ongoing projects addressing sickle cell disease:
- Evaluating the optimal dosing of a drug that can prevent sickle cell obstructions. The drug crizanlizumab has been shown to reduce the rate of painful crises. The team is comparing the efficacy of high dosage and standard dosage in a phase three randomized controlled trial.
- Exploring whether a repurposed Parkinson drug can help replace abnormal haemoglobin with fetal haemoglobin. Babies under four months of age have more fetal hemoglobin—the protein on red blood cells that transports oxygen from mothers to fetus—and experience very few symptoms of sickle cell disease. In the BENEFiTS trial, which is funded by the National Institutes of Health, the team is investigating whether the Parkinson drug benserazide can induce fetal hemoglobin in adults.
- Developing a structured program to improve the transition from pediatric care to adult care. The death rate in young adults with sickle cell disease is seven times higher than other people with the disease. The program was developed in collaboration with SickKids and has helped reduce the number of adolescents that drop out of care, and improved medication and appointment adherence.
- Improving screening for neurological issues that result from sickle cell disease. In collaboration with Drs. David Mikulis at Krembil Research Institute and Joseph Fisher at Toronto General Hospital Research Institute, the team is assessing whether measuring blood flow in the brain when blood vessels are dilated can help predict which patients are at risk for neurological impairment. In parallel, the team is also collaborating with researchers at Centre hospitalier de l'Université de Montréal in Quebec and Hôpitaux Universitaires Henri Mondor in France to develop a rapid and easy-to-administer neurocognitive screening tool to help identify patients at risk of dementia.
“I want to live longer and experience no pain,” says Omar Nelson, an individual with sickle cell disease who is participating in his fifth clinical trial. “I’ve been through a lot and I don’t want anyone to have to go through what I’ve been through. I like to be a part of research studies so they can learn from my experience and help people after me.”
By expanding our understanding of diseases that disproportionately impact people of colour, these projects are a critical step towards providing compassionate care and comprehensive treatments to all.
Support for the crizanlizumab trial comes from Novartis Pharmaceuticals. Support for the BENEFiTS trial comes from National Heart, Lung, and Blood Institute/National Institutes of Health grant 1R33HL147845-01A1. Support for the transition navigator study comes from the Toronto General Hospital Foundation, SickKids Foundation, Novartis and ApoPharma. Support for the neurocognitive screening trial comes from Pfizer.
Dr. Kuo is a member of a committee at Bioverativ and Agios Pharmaceuticals; a consultant for Agios Pharmaceuticals, Alexion Pharmaceuticals, Celgene/BMS, Novartis, Bluebird Bio, Pfizer and Apellis; receives honoraria from Alexion Pharmaceuticals and Novartis; is a scientific collaborator with Abfero and Phoenicia Biosciences; and receives research funding from Pfizer.

Princess Margaret Cancer Centre Clinician Investigators Drs. Richard Ward (L) and Kevin Kuo (R) co-lead the clinical trials at the Red Blood Cell Disorders Program.
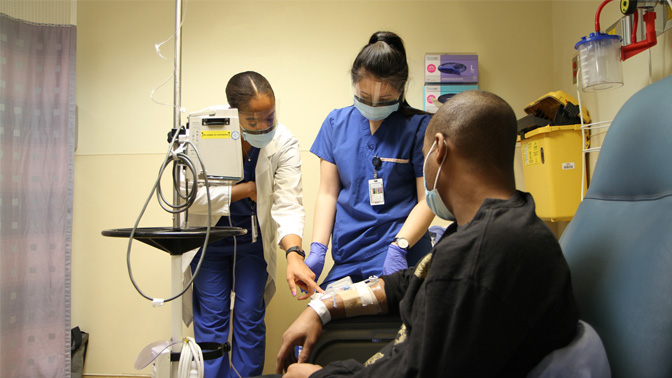
Omar Nelson receives a medication as part of a sickle cell disease clinical trial led by the Red Blood Cell Disorders Program.




